Advanced Capsule Formulations That Deliver Results
A Product Portfolio Built for Performance
Our Products & Capabilities reflect a strong commitment to global quality benchmarks, with all dosage forms manufactured in facilities fully aligned with WHO-GMP standards to ensure safety, consistency, and reliability. Every stage of production is governed by control parameters designed to meet EU-GMP and MHRA expectations, covering key areas such as environmental cleanliness,
equipment hygiene, and contamination prevention. Documentation practices follow structured, internationally accepted formats that support traceability, transparency, and regulatory readiness. All validation activities—including process validation, cleaning validation, and equipment qualification—are performed with strict adherence to global guidelines to guarantee repeatability
and product integrity. Robust QA oversight ensures continuous monitoring of quality attributes, risk management, and compliance throughout the manufacturing lifecycle. With trained personnel, standardized SOPs, and well-maintained infrastructure, we deliver dosage forms that meet therequirements of regulated, semi-regulated, and institutional markets worldwide.
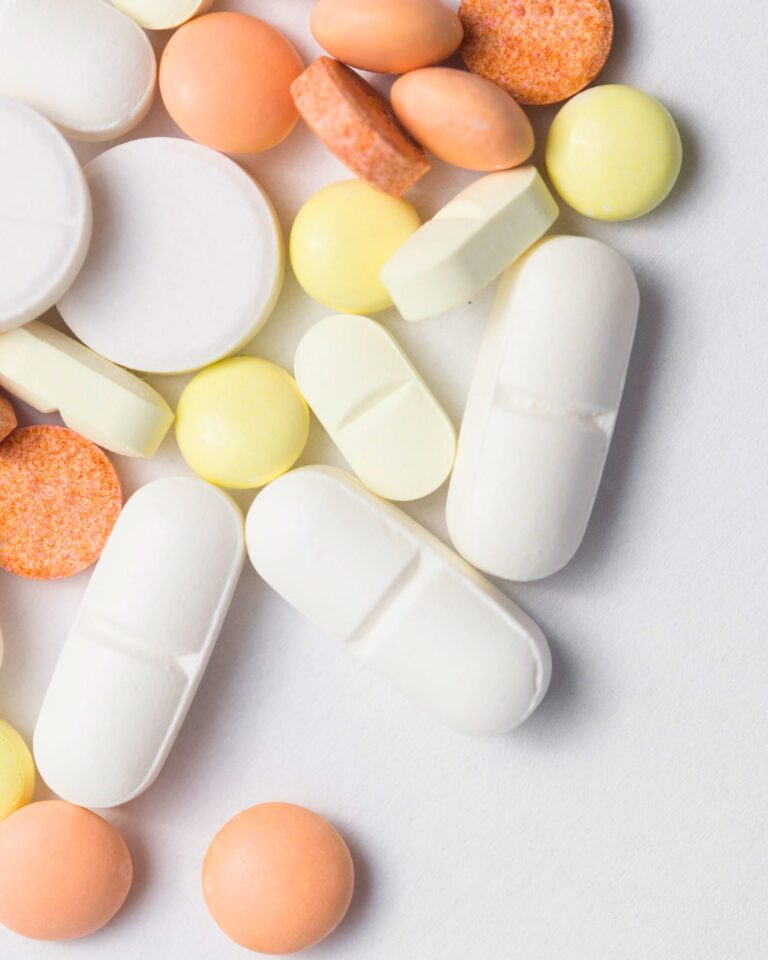

Our Tablets and Capsules manufacturing capability is built on a strong technical framework designed to deliver high-quality, compliant, and consistent pharmaceutical products. All processes strictly follow WHO and EU-GMP validation standards, ensuring each batch meets global expectations for safety, efficacy, and reliability. In-process controls (IPC) are executed as per MHRA aligned SOPs, enabling real-time monitoring of key parameters such as blend uniformity, weight variation, hardness, friability, and dissolution. Our facilities maintain controlled environments for granulation, compression, and encapsulation, preventing cross-contamination and ensuring stable product performance. Granulation suites support both wet and dry granulation with tight control over particle size, flow, and moisture levels. Compression rooms operate under regulated temperature and humidity conditions to protect tablet quality and minimize variability. Encapsulation areas ensure accuracy in fill weight, shell integrity, and capsule locking. All equipment is calibrated and supported by validated cleaning procedures to maintain precision and repeatability. Robust batch documentation ensures full traceability and audit readiness. With skilled teams, standardized procedures, and strong QA oversight, we deliver reliable tablets and capsules aligned with the highest regulatory and market expectations
Our Ointments and Creams manufacturing unit is built on strong quality systems designed to ensure consistency, stability, and global regulatory compliance. Homogenization
processes and emulsion stability are closely monitored in accordance with EU-GMP guidelines, ensuring uniform texture, optimal viscosity, and long-term product performance. Each batch undergoes stringent microbial limits testing aligned with WHO and MHRA expectations to guarantee safety and purity across all semi-solid formulations. Controlled environments, validated equipment, and standardized SOPs support the integrity of every step, from raw material handling to final packaging. Comprehensive in-process checks verify
key quality attributes such as pH, spreadability, and uniformity. Detailed batch documentation and strong QA oversight ensure full traceability and audit readiness. With
trained teams and robust quality controls, our ointments and creams consistently meet international standards for regulated and semi-regulated markets.



Our ORS manufacturing follows a strong technical framework designed to ensure accuracy, stability, and global compliance. Production takes place in dedicated humidity-controlled zones to protect the integrity of electrolytes, prevent lump formation, and ensure consistent powder flow.
Each formulation is prepared using WHO-compliant compositions, guaranteeing the correct balance of glucose and salts for effective rehydration. All documentation follows EU-GMP–aligned stability and IPC formats, ensuring thorough monitoring of critical quality attributes such as blend uniformity, pH, osmolality, and moisture levels. In-process controls are carried out at every stage to maintain precision in mixing, filling, and packaging. Batch records are structured for full traceability and audit readiness, supporting submissions to regulated and semi-regulated markets. With validated equipment, trained personnel, and strong QA oversight, our ORS products consistently meet global
quality and performance requirements.
